The VeBDisease specific project investigates climate-driven changes in vector-borne disease dynamics in Romania and proposes strategies to limit their impact.
VECTOR-BORNE DISEASES AND CLIMATE CHANGE
Climate shapes the transmission, spread, and re-emergence of vector-borne diseases by influencing pathogens, vectors, hosts and human populations. At the same time, climate change transforms ecosystems—including urban areas—in ways that can either support or limit the survival and spread of vectors and their hosts. The vector is an organism (most often an arthropod) that transmits an infectious pathogen from an infected human or animal host to an uninfected target human.
Nowadays it becomes essential to develop an integrated approach in order to understand, anticipate, and fight vector-borne emerging animal and zoonotic infectious diseases. The One Health concept unites human, animal, and ecosystem health into a single, interconnected system.
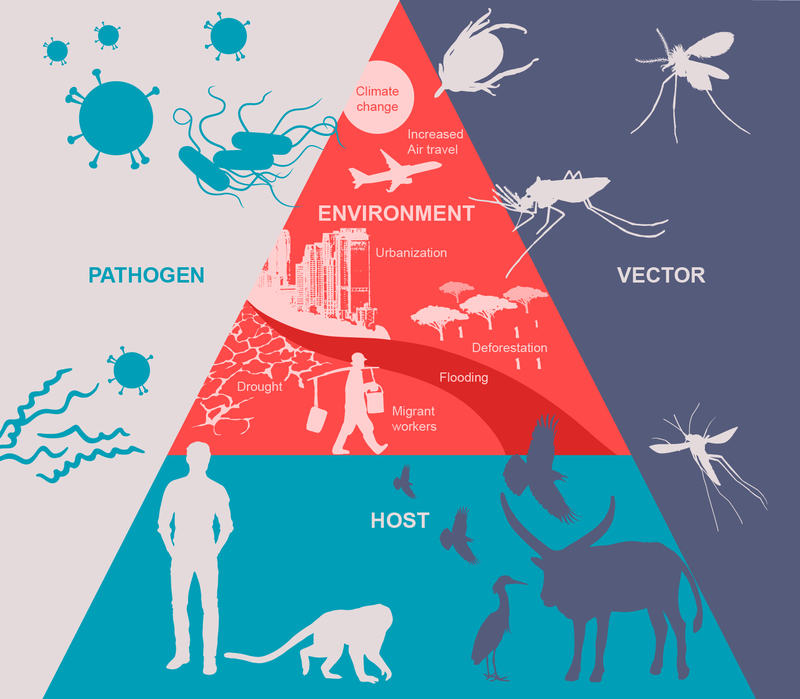
The pathogen–host–vector–environment interface. Image Source: https://doi.org/10.1371/journal.pntd.0008302.g002
DATA AND METHODS
In Europe, several vector-borne viruses pose growing public health concerns, including arboviruses such as chikungunya (CHIKV), dengue (DENV), Zika (ZIKV), Usutu (USUV) and West Nile virus (WNV), alongside tick-borne viruses such as encephalitis (TBEV) and Crimean-Congo hemorrhagic fever virus (CCHFV). For some of them (such as WNV, DENV, and TBEV), several hundred human cases (imported or indigenous) are detected each year in Romania. Considering only the West Nile virus, between 1997 and 2023, more than 900 cases have been confirmed, with an alarming surge between 2017 and 2019.
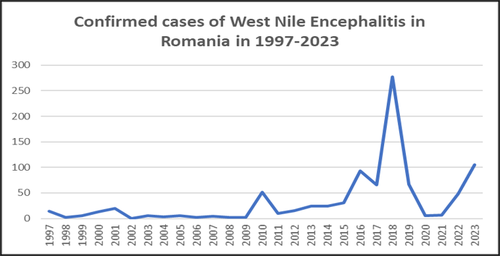
Data Source: National Institute of Public Health, from the WNV Infection Surveillance and Control System
In the context of One Health, an integrated surveillance system needs to be considered - one that combines human and animal health monitoring (detection of cases and identification of animal reservoirs), entomological surveillance (vector identification and risk assessment), and environmental monitoring and modeling.
Activities during the project implementation comprised:
- Vectors testing (mosquitoes, ticks)
- Hosts testing (humans, animals)
- Lab-controlled study on biomanipulated environmental factors (soil microhabitat, soil chemistry and microbiome) for climate-change conditions affecting vectors (mosquitoes and ticks)
- Development of machine learning methods to evaluate risk of vector development based on environmental and meteo variables
CASE STUDIES - Examples:

VECTORS AND HOSTS: Collecting and testing campaigns in TULCEA County (2023-2024)
The Danube Delta region represents an area at risk in the context of climate change for the transmission of infections with CCHFV (Crimean-Congo hemorrhagic fever virus), WNV (West Nile) and TBEV (tick-borne encephalitis). Rural and semi-urban areas with agricultural lands and pastures were chosen. Here, the climatic conditions and diversity of habitats, as well as the presence of domestic and wild animals, support high vector densities and the maintenance of vector populations.
Collected samples were processed in laboratories, with strict quality monitoring. The resulting eluates were numbered for rapid subsequent identification and stored at -80°C until testing by molecular methods. Both sampling campaigns took place from April/May to October/November (periods of maximum vector activity) in 2023 and 2024.
a) Mosquito and tick campaigns

- Mosquito and tick DNA/RNA extractions were performed on pooled samples (50 specimens/sample for mosquitoes and 5–10 specimens/sample for ticks), grouped by species (both), and for ticks, also by developmental stage and feeding status. In total, approximately 3200 mosquitoes and more than 450 ticks were captured.
- As an example, in spring 2024, 1536 mosquitoes belonging to seven species have been identified: Anopheles hyrcanus, Anopheles maculipennis, Culex pipiens complex, Aedes vexans, Aedes caspius, Aedes albopictus, and Uranotaenia unguiculata. MALDI-TOF (Matrix assisted laser desorption and ionisation-Time of flight) mass spectrometry was used for the identification. PureLink Genomic DNA Mini Kit was used for DNA extraction along with the qPCR-CFX96 Real-Time testing instrument. For the analysis, approximately 1507 mosquitoes were tested, from 78 pools.
- Results: WNV virus lineage 2 was identified with mosquitoes captured from Tulcea County.
b) Sheep and cattle blood testing
A total of 1681 blood samples (1059 from sheep and 622 from cattle) were collected during the campaigns run in the two years, 2023–2024.
- Blood samples were taken from clinically healthy animals (sheep and cattle) by venipuncture, using sterile vacutainers with a clot activator, in order to obtain serum. The two species were selected due to their ecological and epidemiological role in the transmission of WNV. As incidental hosts, they develop detectable antibodies after exposure to WNV vectors, even without clinical signs. Their widespread presence and frequent contact with mosquito habitats—particularly Culex species—makes them effective bioindicators of local viral circulation.

- A total of 1316 samples (940 from sheep and 376 from cattle) were tested using the ELISA (Enzyme-Linked Immunosorbent Assay) technique performed to detect antibodies specific to the Flaviviridae family viruses (WNV, USUV, TBEV).
- Results: Preliminary seroprevalences from ELISA have indicated possibly persistent circulation of infected vectors. Positive antibodies were detected in 118 sheep serum samples (12.55%) and 307 in cattle (81.65%). High seropositivity in cattle requires confirmation by seroneutralization (golden-standard) to rule out cross-reactivity and identify the specific virus. Results nonetheless indicate flavivirus circulation in Tulcea County, confirming the role of animals as epidemiological sentinels.

Biomanipulated microenvironment for ticks
In temperate regions like Romania, ticks are a leading cause of vector-borne infections in humans and animals. Among the more than 800 tick species identified worldwide, hard ticks (Ixodidae) are the most diverse and responsible for transmitting diseases such as Lyme disease, TBEV, CCHFV and babesiosis. Their four-stage life cycle (egg, larva, nymph, adult) and the ability to develop off-host make them highly dependent on soil conditions and microhabitats.

Experiments in controlled conditions were performed exploiting the phytotron facility at the Research Institute for Agriculture and Environment, as follows:
- Chemical analysis of soil probes (collected from localities in Tulcea, and more): soil reaction, organic carbon content, grain size/texture analysis
- Environmental simulations and microhabitat variations based on soil samples containing ticks
SOCIETAL IMPACT
Because arthropods and other vectors are ectothermic, higher variations associated with climate change are expected to enhance their abundance, survival, and feeding activity, while accelerating pathogen development within the vector component. Temperature, humidity, and seasonal patterns strongly influence tick and mosquito activity, with milder winters and earlier springs due to climate change potentially extending transmission periods and increasing epidemiological risks. Climate change is already impacting human and animal health—demanding urgent and decisive action.
DELIVERABLES
- Description of work packages (WP1, WP2, WP3, WP4, WP5)
- D5-1.1. Correlation between environmental edaphic factors and developing stages of ticks
- D5-1.3. Prediction of a spreading diseases on human/animals
- D5-1.6. Digital Twin for vector-borne diseases
- D5-3.4. Interdisciplinary review and appraisal of drivers, dynamics and implications of different environmental factors
LIST OF PUBLICATIONS
The VeBDisease website (iuls.ro/dteclimate) contains details on the objectives of this specific project, implementation stages, publication and dissemination.


